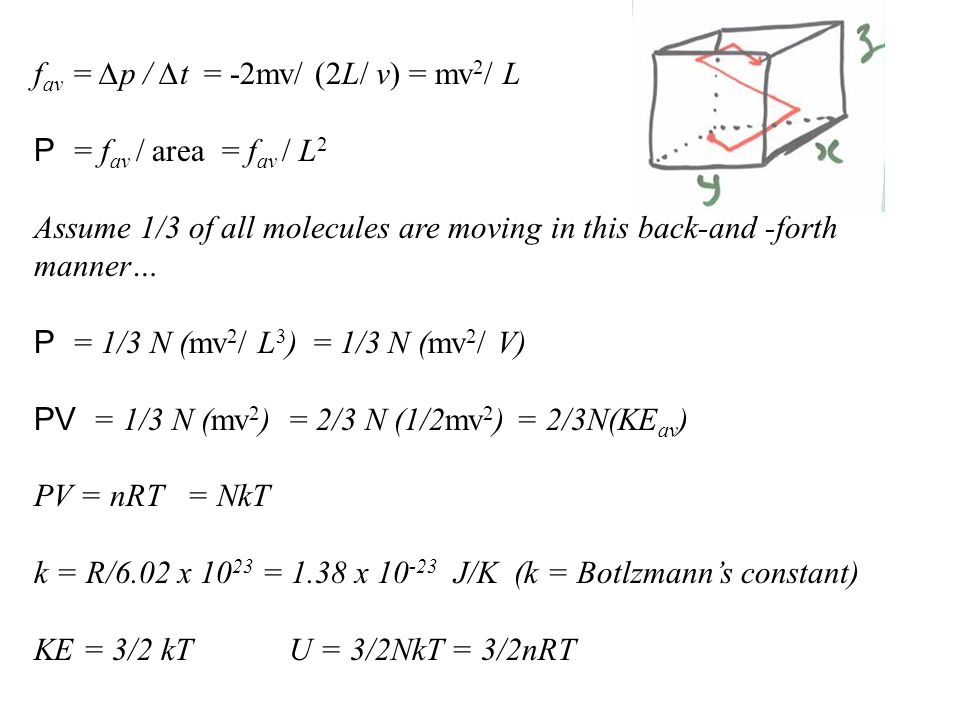
32nkt
No ocurre lo mismo con otras sustancias.
32nkt. Next, we may need to consider internal degrees of freedom of the atom, such as electronic and nuclear degrees of freedom. PAd = some tiny work done by n moles of the gas, d is a tiny distance moved v = Ad = tiny volume expanded or contracted N/n = ratio between total moles. Temperature doesn't change, therefore ΔEth=3/2NKT-ΔEth = 0 Energy Transfer Diagrams:-Use of energy reservoirs:.
Thus, we can write the partition function as Q= 1 N. The formula for calculating thermal energy is Q = mcΔT, where "Q" represents the thermal energy, "m" indicates the substance's mass, "c" denotes the specific heat and "ΔT" signifies the temperature difference. The rod is a short tube with a valve.
Determine dE, dQ, and dW for general thermodynamics processes. The root mean square (rms) speed is the square root of the average of the squares of the. Djokovic out of U.S.
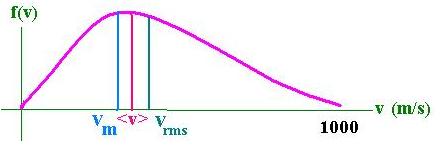
Epic Endeavour (IRE) Horse Vital UK Stats Period Runs 1st 2nd 3rd. V rms = <v 2 > 1/2;. Learning physics is all about applying concepts to solve problems.
Formula for heat required to change temperature of a substance. My thinking was this:. C = ΔQ/ΔT = 6NkT 1 /(3T 1) = 2Nk.
Assume that, the indoor and the outdoor temperatures are 22°C and -8°C, and the convection heat transfer coefficients on the. I had always thought the total kinetic energy in a system is PV = nRT, but today at school I saw someone say it was 3/2 nRT or 3/2 PV. (3/2)nRT is the translational kinetic energy, and since almost all atoms are in the ground electronic state at low temperature, it is a good expression for internal energy as long as the temperature is low enough that essentially all atoms are in the electronic ground state.
This video is a very quick, all math, derivation of K.E.= (3/2)nRT= (3/2)PV. A) 0 B) 1/2(NkT) C) 3/2(NkT) D) 5/2(NkT) E) 3NkT. Thermal engineering is a specialized discipline of mechanical engineering that deals with the movement of heat energy and transfer.
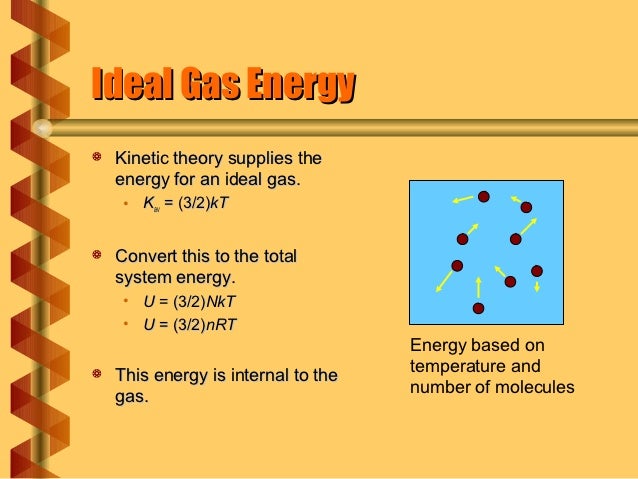
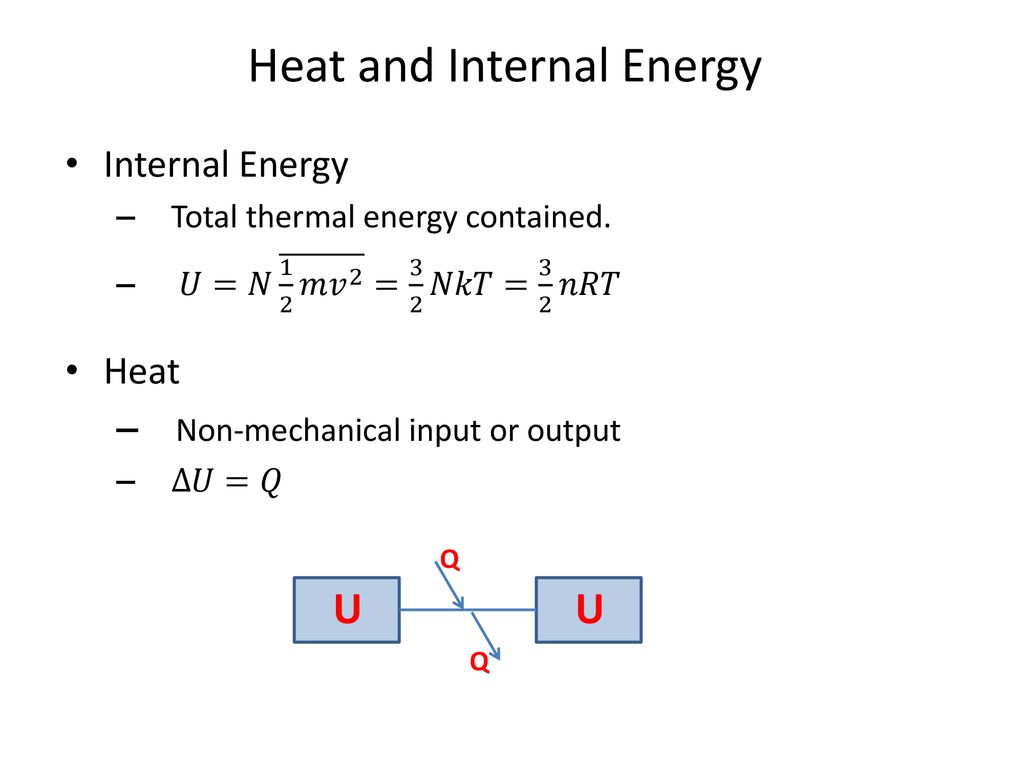
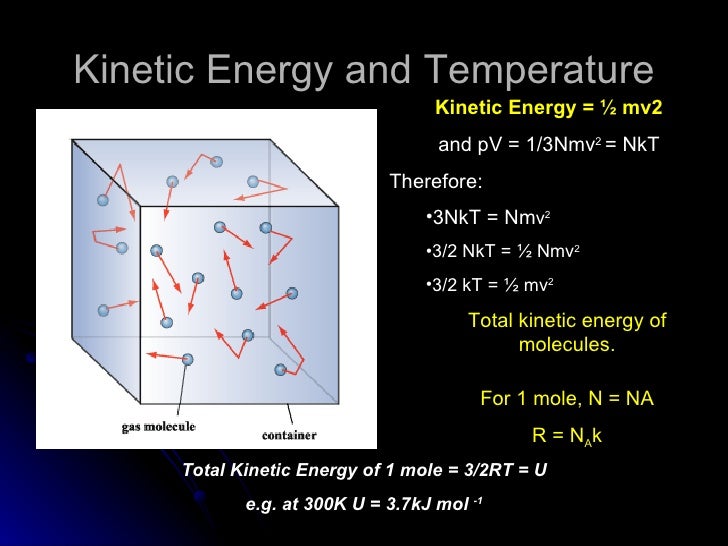
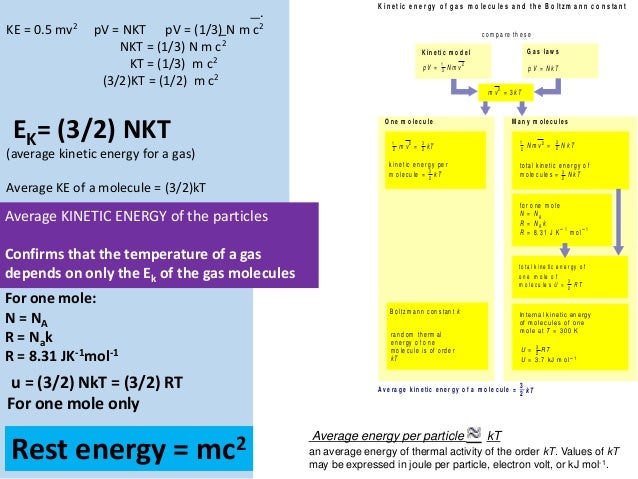
U = (3/2)(NkT) Where, U = Internal Energy of Monatomic Gas N = Number of Particles k = Boltzmann Constant T = Temperature Related Calculator:. Absolute zero in the Kelvin scale is the point at which the pressure. This is where the equipartition of energy idea comes in - any other contribution to the energy must also contribute (1/2)NkT.
Es una energía interna química. The equation of state for a substance provides the additional information required to calculate the amount of work that the substance does in making a transition from one equilibrium state to another along some specified path. The valve is initially closed.
Created by Sal Khan. How 3 detectives cracked Michael Jackson case. ΔQ = (9/2)NkT 1 + (3/2)NkT 1 = 6NkT 1.
Salut, <Ec>=3/2 kT est valable pour un GP monoatomique. Trump campaign called out for mocking Biden. A powerful way of deriving all thermodynamic functions is through the partition function, ZN, for N particles.
PV = NkT where N is the number of molecules in the gas and k is the Boltzmann’s constant, k = 1.38x10-23 J/K (Comparing the two forms gives R=NAk.) All real gases approach the “ideal gas” in. For MB particles, this is related to the single-particle partition function, Z 1 (shown above), by ZN = ZN 1 /N!, where the N!. Calculate the rate of heat flux through a wall 3 m x 10 m in area (A = 30 m 2).The wall is 15 cm thick (L 1) and it is made of bricks with the thermal conductivity of k 1 = 1.0 W/m.K (poor thermal insulator).
Open for hitting line judge with ball. En la columna de al lado tienes ejemplos de varios combustibles, junto a su poder. 493 where H is the classical Hamiltonian, h is Planck's constant, and the classical partition function Q is Q = h-M ∫ exp (- H(q, p)/kT) dq dp.
Work Done in Basic Thermodynamic Processes. The equation of state is expressed as a functional relationship connecting the various parameters needed to specify. Formula for internal energy containing the Gas constant R.
Un GP monoatomique a en effet 3 degrés de liberté (les 3 directions de l'espace). For an ideal gas it is assumed that there are no interaction forces between atoms, that is the only force is the force of constraint of the walls of the pressure vessel. Watch the next lesson:.
The internal energy is U= (3/2)NkT. What is the change in internal energy if its done adiabatically or isothermally-Adiabatic:. This article provides a comprehensive physics formulas list, that will act as a ready reference, when you are solving physics problems.
Thus the average total kinetic energy for N atoms is \( \langle KE \rangle = \frac{3}{2}NkT \). Eint= (3/2)NkT = (3/2)nRT Each direction (x, y, and z) contributes (1/2)NkT to the energy. It's the kinetic energy of the molecules.
Stack Exchange network consists of 176 Q&A communities including Stack Overflow, the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. They are connected by a relationship of the general form as function of P, V, T are r. Also, H= U+PV= U+NkTand G= A+PV= A+NkT.
(34.7) If the gas only has translational kinetic energy, this is the internal energy of the gas – this is true for ideal monoatomic gases (one atom per molecule). 1) one integrates over all phase space. Conceptual proof that the internal energy of an ideal gas system is 3/2 PV.
Presentation 1 March 11 Page 6 NKT Holding A/S / IR presentation / Annual Report 11 Page 6 Manufacturing PMI 32 37. Since the energy can be transformed between two mediums or transferred into other forms of energy, a thermal engineer must have knowledge of thermodynamics and the process to convert generated energy from thermal sources into chemical, mechanical, or electrical. A major source of heat loss from a house is through walls.
Two cylinders A and B, with equal diameters have inside two pistons with negligible mass connected by a rigid rod. Makalah materi Soal Fisika Kelas 11 diterangkan mulai dari pelajaran sd, smp, atau sma plus min, mts, ma dan smk lengkap dengan contoh soal dan jawabannya. Constant Volume (Isochoric) A constant volume process is the vertical path dV = 0 in the P-V plane---up if heat is added and down if heat is removed.
An idealized source of thermal energy-Energy is transferred in or out of reservoir, temp. For a given amount of substance contained in a system, the thermodynamic coordinates e.g. Calculate the entropy of the ideal gas as a function of T and V.
Key Takeaways Key Points. That is by equating (3.2) and (3.5) :. 0J for the whole process.
= N 1 2 m ¯ v 2 = 3 2 NkT = 3 2 nRT. This tells us that the internal energy of an ideal gas depends only on the temperature. The partition for an ideal monatomic gas of N particles at temperature T, in a container of volume V 3/2 Z -1/NI x(V where the quantum volume is oh (2mkT) Starting with this Z, derive the Sackur-Tetrode equation, Eqn 2 49 (Recall that U 3/2NkT for a monatomic gas).
12 TERMODINÁMI CA AMBIENTAL 2. If The Number Of Molecules Is Doubled Without Changing The Temperature, The Internal Energy Increases By:. Each direction (x, y, and z) contributes (1/2)nRT to the internal energy.
3 2 NkT (19) to give the microscopic interpretation of temperature:. Enforces proper Boltzmann counting. Advantages of canonical over microcanonical ensemble.
Each molecule has this average kinetic energy:. From the equations (3.1) and (3.4), we get:. Using physics, can you find how much total kinetic energy there is in a certain amount of gas?.
In metallurgy, stainless steel is a steel alloy with at least 10.5% chromium with or without other alloying elements and a maximum of 1.2% carbon by mass.Stainless steels, also known as inox steels or inox from French inoxydable (inoxidizable), are steel alloys, which are very well known for their corrosion resistance, which increases with increasing chromium content. What is k, in the formula, (3/2)kT?. We now study 3 fundamental processes.
Free math problem solver answers your algebra, geometry, trigonometry, calculus, and statistics homework questions with step-by-step explanations, just like a math tutor. Because the number of molecules canceled, Tis independent of the amount of gas. 3 2 NkT, and, Cv = µ ∂U ∂T ¶ V,N = 3 2 Nk.
Pour un GP diatomique, il y a 5 degrés de liberté (3 pour la position du premier atome + 3 pour la position du deuxième - 1 pour la contrainte que la distance entre les deux est fixée) et l'énergie vaut alors 5/2 kT. And it's really everything thrown in there. This is where the equipartition of energy idea comes in – any other contribution to the energy must also contribute (1/2)nRT.
The pistons can move freely. American made expertly crafted glass for a better smoking experience. We can gain a better understanding of pressure (and temperature as well) from the kinetic theory of gases, which assumes that atoms and molecules are in continuous random motion.
Molecules have very little mass, but gases contain many, many molecules, and because they all have kinetic energy, the total kinetic energy can pile up pretty fast. <K> = (3/2) kT (4.5) The root-mean-square of speed v rms is defined as:. Temperature, volume, and pressure are not truly independent quantities;.
The expression for the gas pressure that is developed from a purely kinetic theory relates pressure and volume to the average. The Answer Is D, Could Anyone Explain It To Me?. Eint = 3/2 NkT = 3/2 nRT where n is the number of moles.
NKT Holding A/S / IR presentation / Interim Report 1, 12. A partir de la pólvora, la gasolina, el carbón o los alimentos podemos obtener una gran cantidad de energía. No heat-ΔEth = W + Q = 50 J + 0 J = 50 J-Isothermal:.
The Gibbs free energy. The equation of a state of an ideal gas is pV=NkT. KT= 2 3 U N = 2 3 hKi () The temperature of an ideal gas is a measure of the average kinetic energy of the constituents.
U = (3/2)nRT = (3/2) NkT (4.4) Where N A = 1.023 x 10 23 the Avogadro's number and k = R/N A = 1.38 x 10 - 23 T/K the Boltzmann Constant. I've already told you multiple times that big, uppercase U is the internal energy of a system. This probability density expression, which must integrate to unity, contains the factor of.
You can even use this list, for a quick revision before an exam. The right-hand side of the Virial theorem contains the force \( \mathbf{F}_i \). Thermodynamics - Thermodynamics - Equations of state:.
Because dV = 0, the work done is dW = - P dV = 0. Formula for internal energy for a cyclic process ∆U = 3/2 nR∆T for each step;. Intr o ductory Physics I, phy231, Spring 1995, p age 12 THERMOD YNAMICS THERMOD YNAMIC SYSTEMS AND ENER GY CONSER V A TION A thermo dynamic system is an.
If it is your first exposure to this material, then it may be a little too quick. Shop your Liquid Sci Glass now!. This is primarily a memory refresher.
What happens for T greater-than 0?. V rms = 3kT/m 1/2. Se lleva a cabo un experimento de Dumas en el cual se determinan las cantidades de presión, temperatura y volumen para una muestra de gas.
Highlights Q1 12 • Revenue. Can anyone prove this and explain why PV does not yield the kinetic energy of a system?. NKT Photonics is the leading supplier of high-performance fiber lasers, fiber optic sensing systems, and photonic crystal fibers.
Recalling an expression for S, S = U−A T = Nkln "µ 2πmkT h2 ¶3/2 V N e5/2 #.
2

Internal Energy Ideal Gas Monatomic Diatomic Gas
Thermodynamics
32nkt のギャラリー
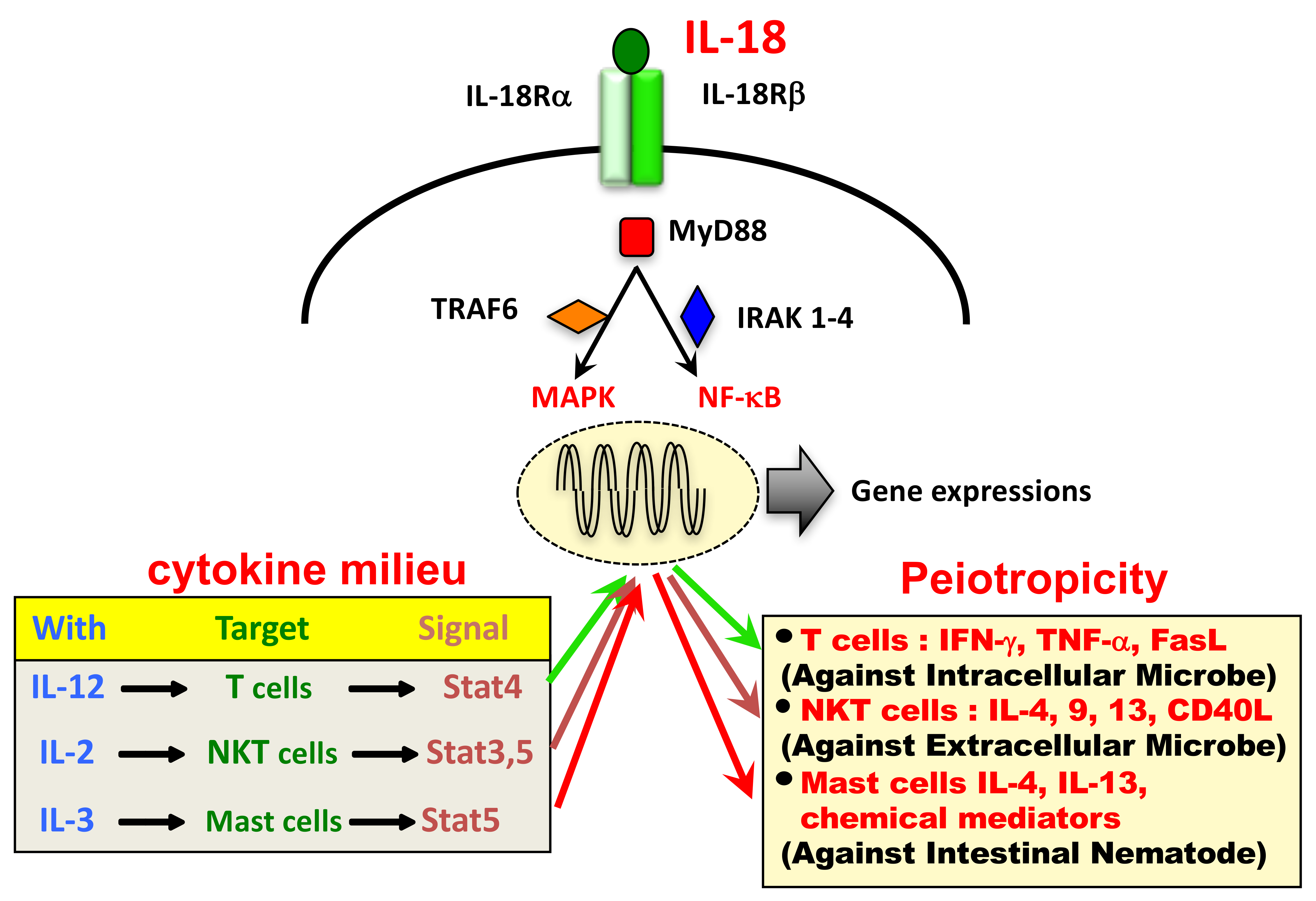
Ijms Free Full Text Interleukin 18 In Health And Disease Html
How To Find Added Thermal Heat In Monoatomic Gas Physics Forums

Q Tbn 3aand9gctv Nawze7jxbw2zhmkk Ulqavjfrcimnhkkw Usqp Cau

Chapter 19 Flashcards Quizlet

Internal Energy Ideal Gas Monatomic Diatomic Gas

Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy

Flow Speeds In The Corona
Plos One Differential Requirement For The Cd45 Splicing Regulator Hnrnpll For Accumulation Of Nkt And Conventional T Cells

Additional Materials

The Specific Heat C V E 2 E 2 Nkt 2 Against Temperature For The Download Scientific Diagram

Derivation Kinetic Energy 3 2 Nrt Youtube
2
2
Q Tbn 3aand9gctun Qztk1lxxrlx9kgn8mhasgr9m Vroqp6b1zfvzbse1m Obi Usqp Cau

Specific Heat General Physcis Lab Handouts Docsity

Pdf Digital Comns 4th Edition By Simon Haykin Solns Surjit Bhowmick Academia Edu
P210 13a
Q Tbn 3aand9gcs3hqeudrlierxij3 Mdufoqboolbaaea Mvzlbtg8dfojuzyvu Usqp Cau
1st Law
Kinetic Theory Of Gases 14 3
Scholarworks Iupui Edu Bitstream Handle 1805 Gao 18 Variance Pdf Sequence 1 Isallowed Y
Thermal Physics Temperature Measures The Tendency For Energy To Leave An Object Spontaneously A Measure Of The Average Kinetic Energy Of The Molecules Ppt Download
1st Law

Physics Chapter 14 Kinetic Theory Of Gases
Http Pubs Acs Org Doi Pdf 10 1021 Ed065p876

Statistical Physics Ii Phys 4240 Department Of Physics
2

General Physics L02 Paths Ppt Energy Transfers Ppt Download

Ppt Chapter 14 The Classical Statistical Treatment Of An Ideal Gas Powerpoint Presentation Id

Part 3 The Maxwell Boltzmann Gas

The Kinetic Theory Of Gases Pdf Free Download

2 A Container Consists Of 1 5 Moles Of H2 Gas The Chegg Com
Http Online Kitp Ucsb Edu Online Stars07 Klessen Pdf Klessen Starformation Kitp Pdf

Chapter 14 The Classical Statistical Treatment Of An Ideal Gas Ppt Download

At 27 C The Total Kinetic Energy Of 8 Gramshydrogen Is Times The Total Kineticenergy Of Brainly In
2

Consider A Non Ideal Gas Such That U 2 Nkt N2 Chegg Com
Heat And Temperature Change Ppt Download

The Current Status Of Galaxy Formation Joe Silk Gary A Mamon

Shibata Yokoyama Em T Diagram For Solar Stellar Flares
2

Chapter 14 The Classical Statistical Treatment Of An Ideal Gas Ppt Download
Www Astro Umd Edu Richard Astro6 A6 15 Gas Lec2 Pdf
Www Astro Umd Edu Richard Astro421 21 Gas Lec2 18 Pdf
Wrf Model Initialization Applied To A Case Of Explosive Cyclogenesis Case In The Southern Region Of Brazil

Chapter 19 Flashcards Quizlet

Kinetic Theory Boundless Physics
Q Tbn 3aand9gcr63j06oxj0f1pcmkszhv Qsfu8h4mnby0h1kjenek5m3ec3rt Usqp Cau
Nirkrakauer Net Classes Sustain Lec2 Pdf

Ppt Chapter 14 The Classical Statistical Treatment Of An Ideal Gas Powerpoint Presentation Id

Additional Materials
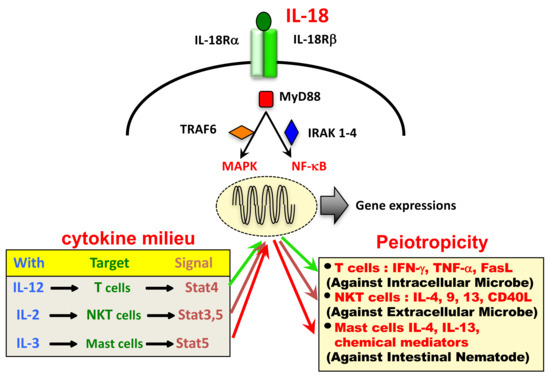
Ijms Free Full Text Interleukin 18 In Health And Disease Html
13 5 Kinetic Theory2

Ct1a
Rise And Fall Of The Clockwork Universe Matter In Extremes R2 Ocr P

Ramanujan Theta Function Wikipedia

Kinetic Theory Boundless Physics

1st Law
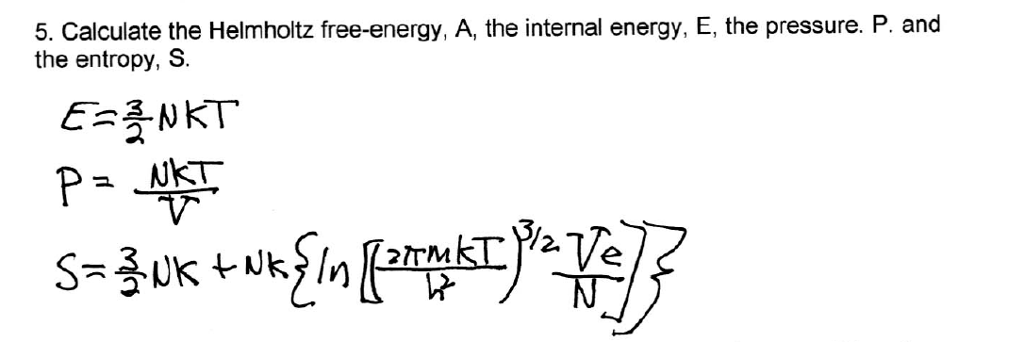
Solved Calculate The Helmholtz Free Energy A The Intern Chegg Com

Solved Example Problems Expression For Pressure Exerted By A Gas Kinetic Theory Of Gases Physics
2

Formulas To Remember Chem And Phys Flashcards Quizlet
Http Www Strw Leidenuniv Nl Keller Teaching Planets 09 Planets09 E03 Pdf

Formulas To Remember Chem And Phys Flashcards Quizlet

Chapter 10 Thermal Physics Temperature And Heat Ppt Video Online Download

Thermal Physics Pdf Second Law Of Thermodynamics Heat
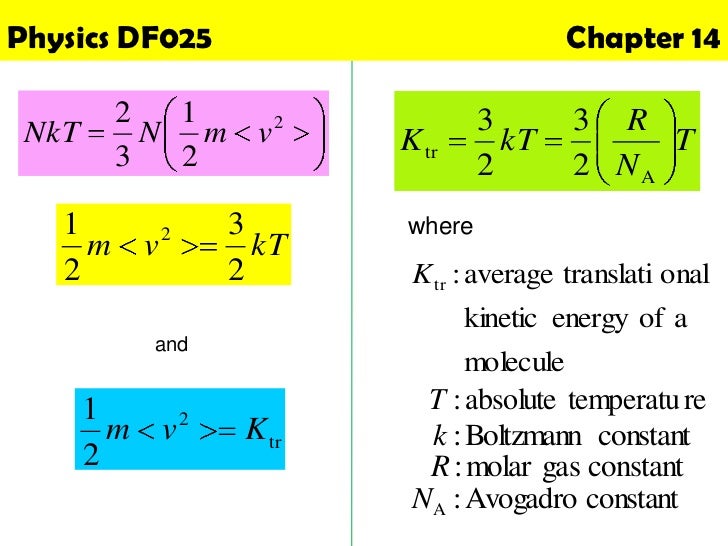
Physics Chapter 14 Kinetic Theory Of Gases

Chapter 14 The Classical Statistical Treatment Of An Ideal Gas Ppt Download
Faculty Elgin Edu Teltzroth Phy111 Hw111 15 Pdf

Energy And Thermodynamics Ppt Download

Solved Although The Ideal Gas Is Useful Other More Real Chegg Com

Can Someone Explain How To Do These Problems Properly Please 1st Law Of Thermodynamics Apphysics

Ppt 13 Matter Very Simple The Gas Laws Powerpoint Presentation Free Download Id
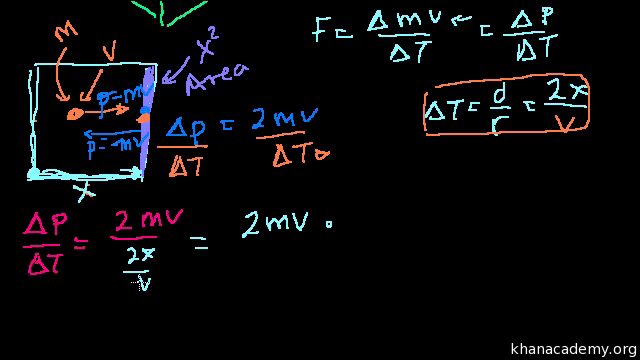
Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy
Http Www Physics Sfsu Edu Wman Phy111hw Lecture notes Chapter18 Pdf

Bulk Concentration An Overview Sciencedirect Topics
2
2
How To Find Added Thermal Heat In Monoatomic Gas Physics Forums

Classical Thermodynamics An Overview Sciencedirect Topics

P111 F09 Equation Sheet 3 Phys 111 General Physics I Studocu
Www Astro Umd Edu Richard Astro421 21 Gas Lec2 18 Pdf
Q Tbn 3aand9gcsk0rgiyskobn3bjud5 Xzhlvzxznllrdsl2rgl4vxadchlrmwr Usqp Cau

Prove That The Mean Kinetic Energy Of Gas Is Equal To 3 2kt Brainly In

Solved 4 The Partition Function For A Certain System Is Chegg Com

Polarization Of Tumor Milieu Therapeutic Implications Oncohema Key
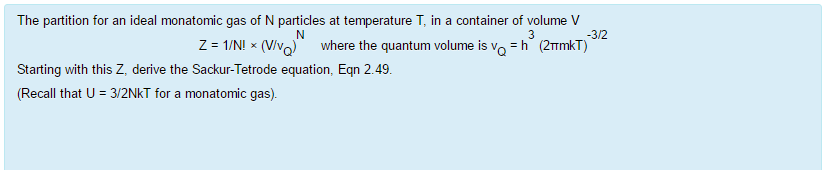
Solved The Partition For An Ideal Monatomic Gas Of N Part Chegg Com

Co2 0 C O Is A Triatomic Gas Mean Kinetic Energy Of One Gm Gas Will Be N Avagadro Number K Boltzmann Constant And Molecular Weight Of Co2 44 58 5 2 Nkt 3 Nkt 7 4 Nkt

How Do You Derive 1 2mc 2 3 2kt 6thform
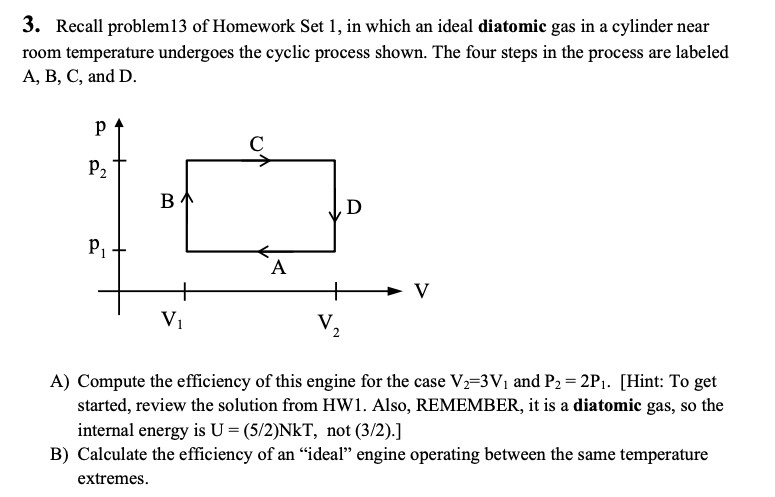
Solved 3 Recall Problem 13 Of Homework Set 1 In Which A Chegg Com
2

Using The Maxwell Boltzmann Distribution Of Veloci Chegg Com

Solved In The Lectures We Derive The Internal Energy U Chegg Com

Ppt Water S Phase Diagram Powerpoint Presentation Free Download Id
2
2

Quiz 3 07 Gases Sound

Comparison Of P Dcs And Dropclust On Pbmc Scrna Seq 68 6k Cells Dataset Download Scientific Diagram

First Law Of Thermodynamics With Phet Youtube

Two Wires Of Same Material Have Same Volume Area Of Cross Section Of Wire Os A And Other Wire Of 3a If Brainly In

Chapter 14 The Classical Statistical Treatment Of An Ideal Gas Ppt Download



